Pharmaceutical Quality Systems
Pharmaceutical or medical product and cause contamination In such cases, systems, equipment and Generation of clean steam relies on the evaporation of good quality feedwater Contaminants remain in the concentrated feedwater within the generator, and are periodically removed by “blowdown”.

Pharmaceutical quality systems. A harmonized pharmaceutical quality system applicable across the lifecycle of the product emphasizing an integrated approach Q10 Pharmaceutical Quality Systems Q8, 9, & 10 Quality by Design, Risk Management, and PQS provide greater product assurance of quality 22. Pharmaceutical Companies This page contains corporate information for pharmaceutical companies marketing products in the United States Information includes company addresses, telephone numbers, stock quotes, links to corporate websites, lists of medicines, support and employment opportunities where applicable. For the 21st century – there has been a growing awareness for the significance of the quality of the pharmaceutical products (Woodcock, 04).
CAPA within the Pharmaceutical Quality System 1 Martin VanTrieste, RPh SVP Amgen ICH Q10 Conference October 46, 11 – Arlington, Virginia November 1416, 11 – Brussels, Belgium. In the pharmaceutical industry, the most common quality KPI was rejected batches followed by the number of complaints, product defects, and deviations The number of complaints was the most common. 1 Introduction The quality in the pharmaceutical industry has become a very important topic Since the world has gathered together to harmonize its practices and guides and the launching of the FDA current good manufacturing practices – the cGMP;.
The pharmaceutical quality control laboratory serves one of the most important functions in pharmaceutical production and control Most manufacturers use systems that provide for the. Heat and Control strategic partner CEIA, the world leading innovator of industrial metal detection systems, has released new pharmaceutical metal detection systems to further enhance inspection and quality control Ensuring pharmaceutical, nutraceutical and healthcare manufacturers have the. 11 This guide is applicable to all elements of pharmaceutical and biopharmaceutical manufacturing systems including good manufacturing practice (GMP) utility equipment, process equipment, supporting utilities, associated process monitoring and control systems, and automation systems that have the potential to affect product quality and.
For the 21st century – there has been a growing awareness for the significance of the quality of the pharmaceutical products (Woodcock, 04). This document aims to give guidance to pharmaceutical manufacturers and inspectors of pharmaceutical manufacturing facilities on the design, installation, qualifi cation and maintenance of the HVAC systems These guidelines are intended to complement those provided in Good manufacturing practices for pharmaceutical products (1) and should be read. Pdf (247 KB) White Paper Abuse Deterrence for the Pharmaceutical Market pdf (194 MB) White Paper Surface Analysis Exposes Counterfeit Medicines pdf (462 KB) White Paper Single Use Systems in the Pharmaceutical Industry Advantages.
Heat and Control strategic partner CEIA, the world leading innovator of industrial metal detection systems, has released new pharmaceutical metal detection systems to further enhance inspection and quality control Ensuring pharmaceutical, nutraceutical and healthcare manufacturers have the. Seven Critical Concepts of the FDA’s Quality Systems Guidance FDA Issues Final Guidance on Quality Systems Approach From the discovery of penicillin in the late 19s to the breakthrough anticholesterol and antiHIV drugs being manufactured today, the pharmaceutical industry has always been in the front line of developing new technologies. A pharmaceutical quality hazard or to reduce it to an acceptable level 290 design qualifi cation (DQ) Documented evidence that the premises, supporting systems, utilities, equipment and processes have been designed in accordance with the requirements of good manufacturing practices (GMP).
11 This guide is applicable to all elements of pharmaceutical and biopharmaceutical manufacturing systems including good manufacturing practice (GMP) utility equipment, process equipment, supporting utilities, associated process monitoring and control systems, and automation systems that have the potential to affect product quality and. Pharmaceutical Quality/Manufacturing Standards (CGMP) This guidance is intended to help manufacturers implementing modern quality systems and risk management approaches to meet the requirements of. Quality by Design for the Pharmaceutical Industry pdf (137 KB) White Paper Are you Ready for USP 232>/233>?.
2 General principles for pharmaceutical water systems 21 Pharmaceutical water production, storage and distribution systems should be designed, installed, commissioned, quali"ed and maintained to ensure the reliable production of water of an appropriate quality It is necessary to validate the water. Pharmaceutical or medical product and cause contamination In such cases, systems, equipment and Generation of clean steam relies on the evaporation of good quality feedwater Contaminants remain in the concentrated feedwater within the generator, and are periodically removed by “blowdown”. The pharmaceutical quality system can normally be evaluated during a regulatory inspection at the manufacturing site Potential opportunities to enhance science and riskbased regulatory.
Seven Critical Concepts of the FDA’s Quality Systems Guidance FDA Issues Final Guidance on Quality Systems Approach From the discovery of penicillin in the late 19s to the breakthrough anticholesterol and antiHIV drugs being manufactured today, the pharmaceutical industry has always been in the front line of developing new technologies. With six manufacturing facilities already certified by EXCiPACT, Ashland is well on its way to have all pharmaceutical excipient facilities certified by the end of 17 Ashland continues its quest for high quality, which in turn, simplifies and streamlines your sourcing options.

Quality Assurance Pharmaceutical Quality Systems In Making Medicines

Ich Q10 Where Gmp And Good Business Practice Meet
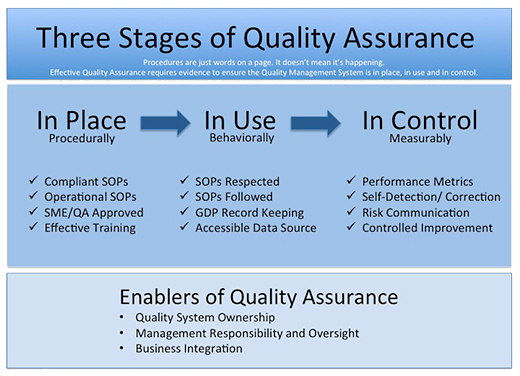
Three Stages Of Quality Management System Implementation And Oversight Quality Digest
Pharmaceutical Quality Systems のギャラリー

What Are The Stages Of Implementing A Quality System In Pharma Manufacturing Uality System In Pharma Manufacturing Pharmaceutical Manufacturing Pharma System

Ppt Quality Management And Pharmaceutical Quality System Powerpoint Presentation Id
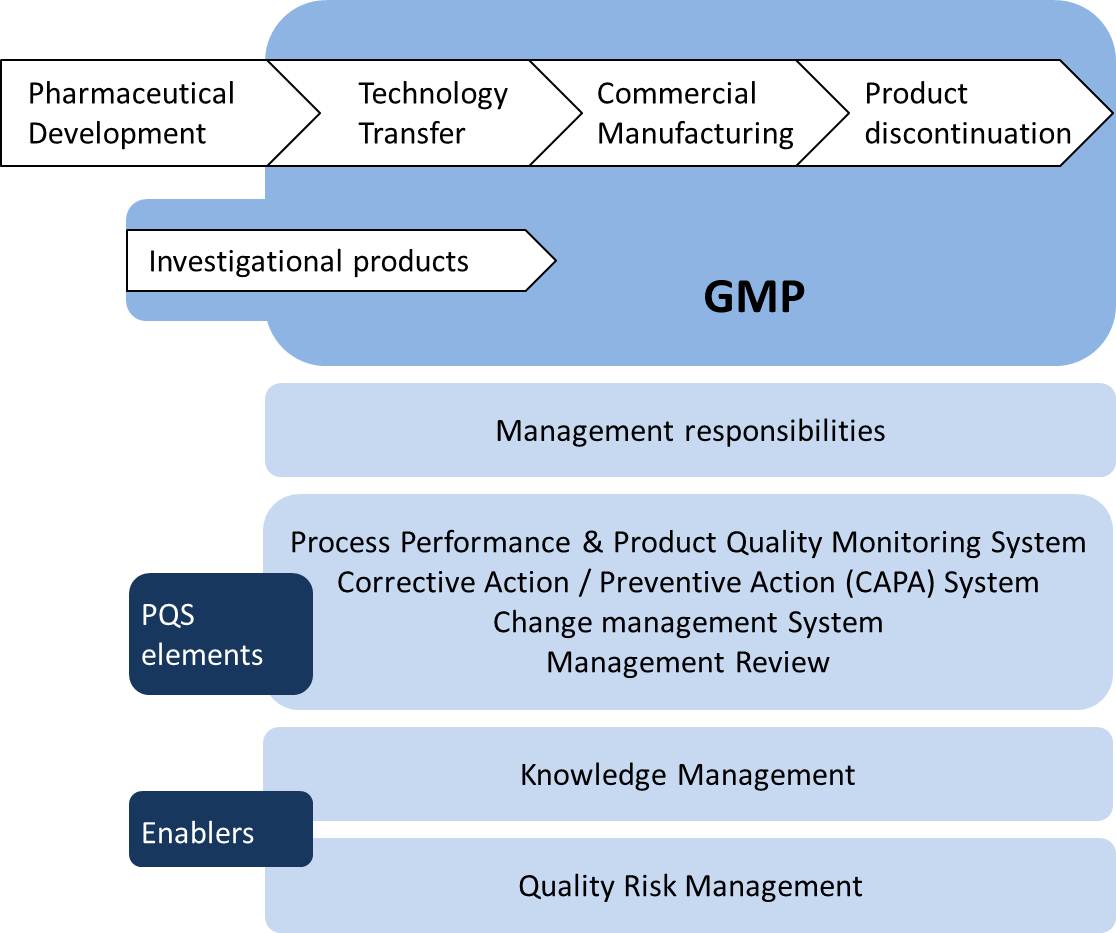
Pharmaceutical And Vaccine Quality Illustrated

Six System Inspection Approach Source Fda Pharmaceutical Gmp Download Scientific Diagram

Pharmaceutical Quality Systems Ich Q10 Conference Business Case For Quality Pdf Free Download
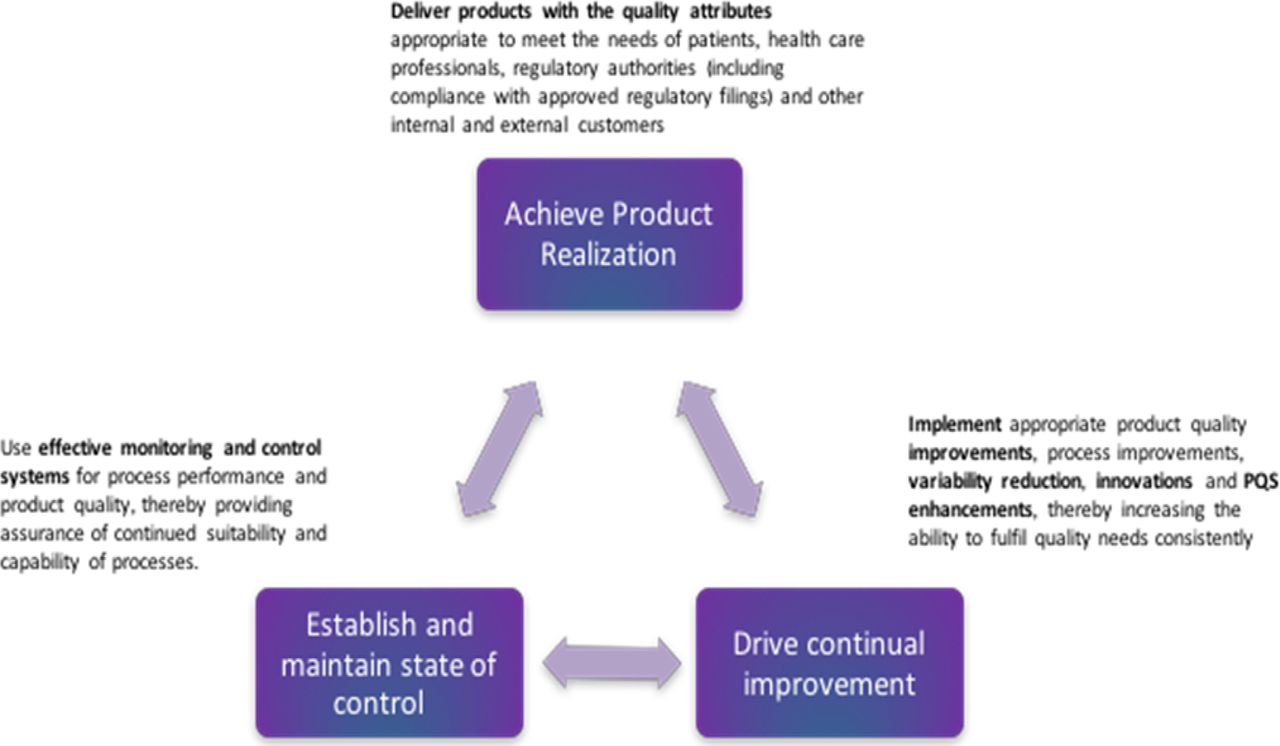
Demonstrating Pharmaceutical Quality System Effectiveness And Driving Continual Improvement Evidence Based Risk Reduction Pda Journal Of Pharmaceutical Science And Technology

Design And Implementation Of Quality Pharmaceutical Systems

Pharmaceutical Quality System Pqs Lundsberg Consulting

Your Free Pharmaceutical Quality System Health Check

Quality Management Systems Training Course Ispe International Society For Pharmaceutical Engineering

Calameo Fda Guidance The Quality Systems Approach To The Pharmaceutical Good Manufacturing Practices

How To Gauge Your Quality Management Technology Maturity
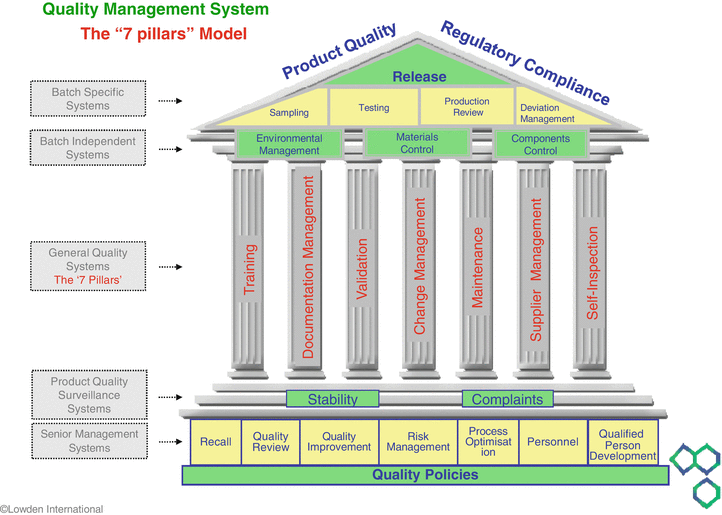
Pharmaceutical Quality Systems Springerlink

Pharmaceutical Contract Manufacturing Organization Overview

Low Code Qms Software For Pharmaceuticals 10x Faster Deployment

Ich Q10 Pharmaceutical Quality System
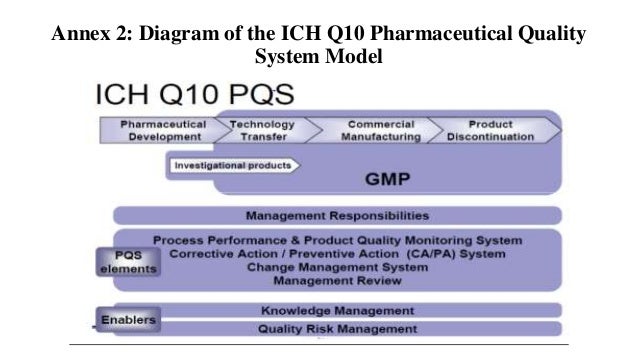
Pharmaceutical Quality System

The Future Of Quality Control

Pharmaceutical Quality Systems Pqs 101 A Managers Best Friend

Pharmaceutical Quality Systems 1st Edition Oliver Schmidt Routl

Commissioning And Qualification Verification In The Pharmaceutical Product Process Lifecycle Quality Systems

Is Gmp Quality System Auditing Fundamentally Flawed A Data Integrity Alternative

Good Manufacturing Practices Cgmp When Do They Apply Advarra

Pharmaceutical Quality System

A Process Approach To Pharmaceutical Quality Systems A Guide To Ich Q10 Compliance
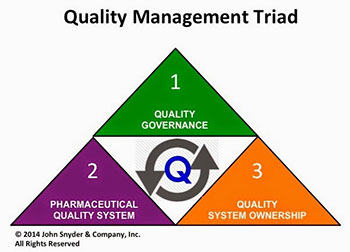
Essentials For Sustaining An Operational Pharmaceutical Quality System Quality Digest

Delivering A Modern Effective Pharmaceutical Quality System Ppt Download

Compliance By Design For Pharmaceutical Quality Control Laboratories Insight From Fda Warning Letters Pdf Download M A N O X B L O G

Niper Hyderabad Workshop On Pharmaceutical Quality Assurance And Quality Control Qa Qc In Collaboration With Pharmexcil Nib Noida On 11 13 Oct Module 1 18 Oct Module Ii 19 Mansukhmandviya Dvsadanandgowda Rajneeshtingal

International Council Of Harmonization Q7 Q14 Investigations Of A Dog

Gmp International Master Reference Guide

Dynamics Of Pharmaceutical Quality Systems For The Export Of

Incorporating Data Integrity Into Your Pharmaceutical Quality Management System

Quality System Model Ich Q10 Ppt Video Online Download

Ppt Requirements For The Quality Of Api From Fda Perspective Powerpoint Presentation Id

Pharmaceutical Quality Systems Springerlink

10 Key Ingredients For Small Pharma Gcp Quality Systems

Gamp In Pharmaceutical Quality System An Overview Youtube

Good Manufacturing Practices For Pharmaceuticals Seventh Edition Drugs And The Pharmaceutical Sciences Kindle Edition By Bunn Graham P Professional Technical Kindle Ebooks Amazon Com

Api For Human Use Api For Veterinary Use Pharmaceutical Intermediates Animal Health Drugs Changzhou Yabang Qh Pharmachem Co Ltd
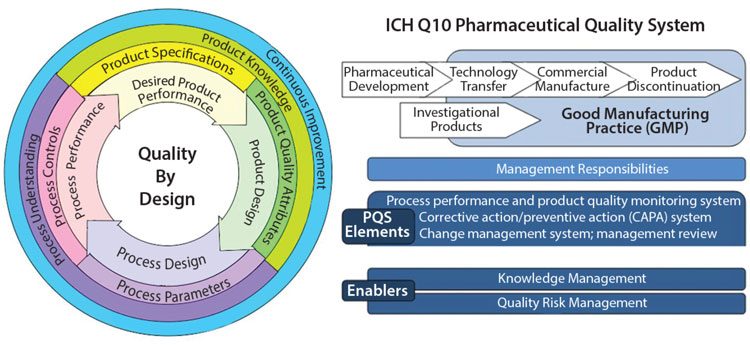
Quality Assurance Pharmaceutical Quality Systems In Making Medicines

Live From Pda Fda Fda Considers Incentivizing Quality Management Maturity Qmm Healthcare Packaging

Change Control Procedure In Pharmaceuticals Quality Assurance

Ich Q10 The Ich S Pharmaceutical Quality System Project Management Templates Research Proposal Example Pharmaceutical

How To Achieve Quality By Design Qbd In A New Pharmaceutical Fill Line Cleaning And Disinfection Series Part 3 Of 6 Particle Measuring Systems

How Digital Solutions Enhance Pharma Quality Systems Amplelogic

What Is Pharma 4 0 Insights Into The Paradigm Shift

Webinar Pharmaceutical Quality Systems Pharma Biotech Youtube

Figure 1 From Short Review On Quality By Design A New Era Of Pharmaceutical Drugdevelopment Semantic Scholar

Pharmaceutical Quality Systems Contract Pharma
Blog The Fda Group Quality Systems
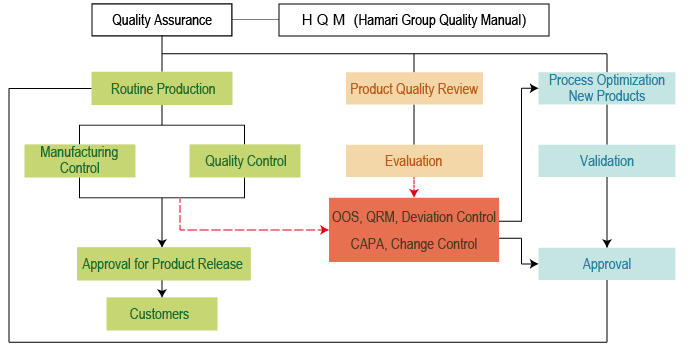
Quality Policy And Advanced Quality Assurance System Hamari Chemicals Ltd

Steps For Implementing A Quality Management System The Successful Way

Ich Q10 Pharmaceutical Quality System
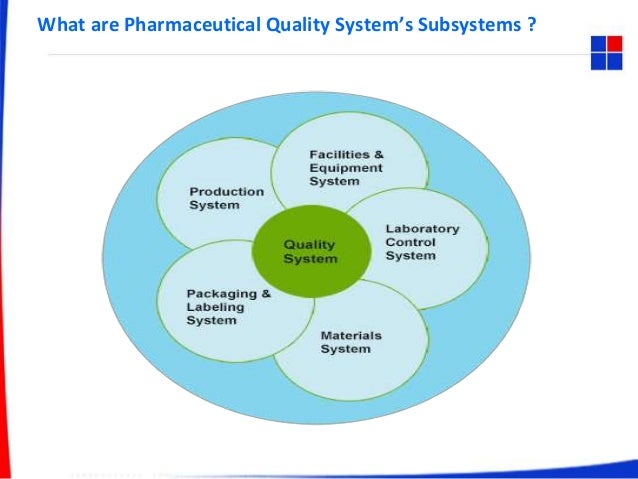
What Are Pharmaceutical Quality System S Subsystems
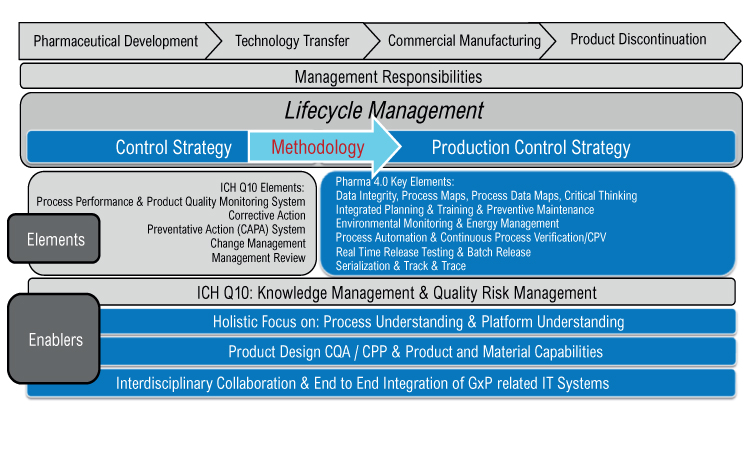
A Holistic Approach To Production Control Pharmaceutical Engineering

Quality Assurance Pharmaceutical Quality Systems In Making Medicines
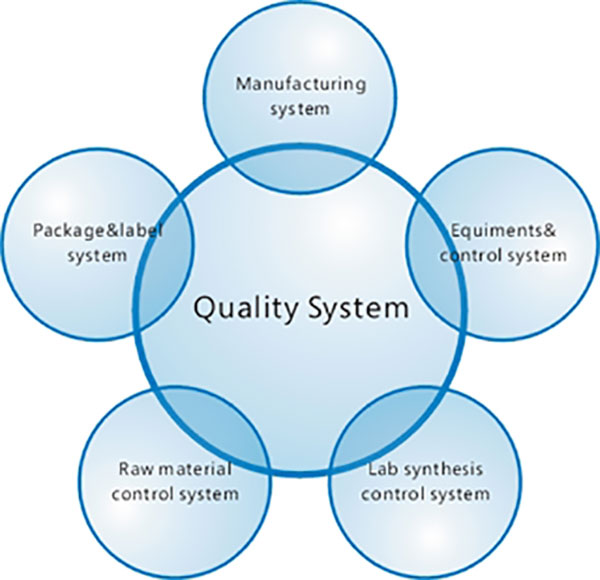
Welcome To Lepharm

Pharmaceutical Quality System Pqs

Basic Principles Of Pharmaceutical Quality System Pqs Ich Q10 Ppt Download

How To Achieve Quality By Design Qbd In A New Pharmaceutical Fill Line Set Up And Process Interventions Series Part 4 Of 6 Particle Measuring Systems

How Does A Company Demonstrate The Implementation Of Pqs In Accordance With Ich New Drug Approvals

7 Critical Fda Concepts For Pharmaceuticals Quality Systems Gxp Lifeline Food And Drug Administration Quality Management System

Nsf Pharma Technology Focus Issue 80 March 19
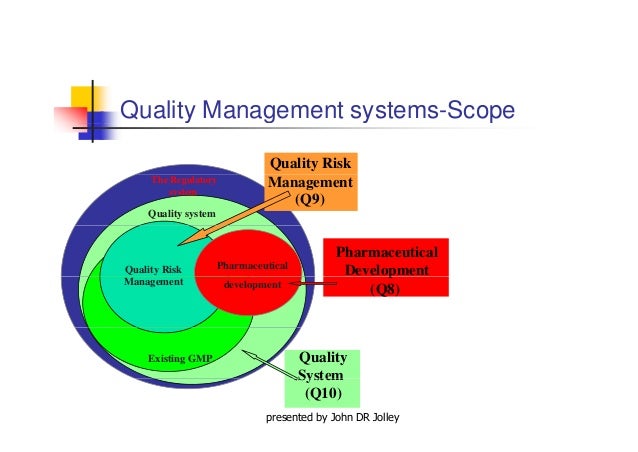
Industry Implications Of Pharmaceutical Quality Ich Guidelines

Pharmaceutical Quality Control Rough Draft Authorstream

The Ways Pharmaceutical Quality Systems Will Benefit Your Company By Johnathonp Issuu

Pqs Pharmaceutical Quality Systems By Acronymsandslang Com
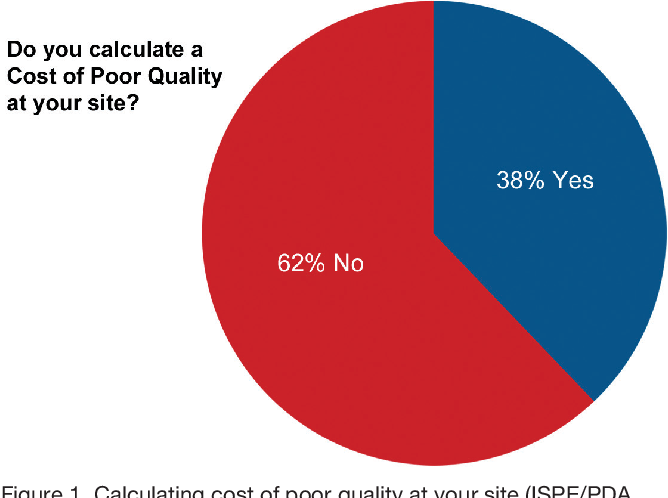
Pdf Quality Systems Pharmaceutical Quality Management Moving From Quality Control To Quality Assurance By Guy Wingate Semantic Scholar

Pharmaceutical Quality System

Pharmaceutical Quality System Q10 Pdf Download M A N O X B L O G
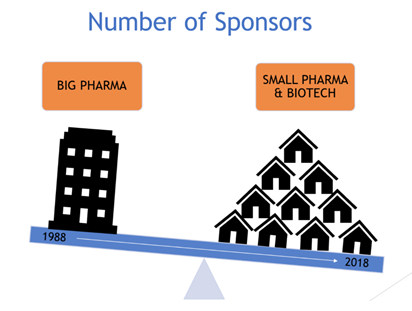
10 Key Ingredients For Small Pharma Gcp Quality Systems

Quality Metrics For Pharmaceutical Manufacturing Pharmaceutical Guidelines

Quality Systems And Controls For Pharmaceuticals Medicine Health Science Books Amazon Com

How Pharmaceutical Quality Systems Will Benefit Your Company By Resolsystems Com Issuu

What S The Most Effective Pharmaceutical Quality Management System In 19
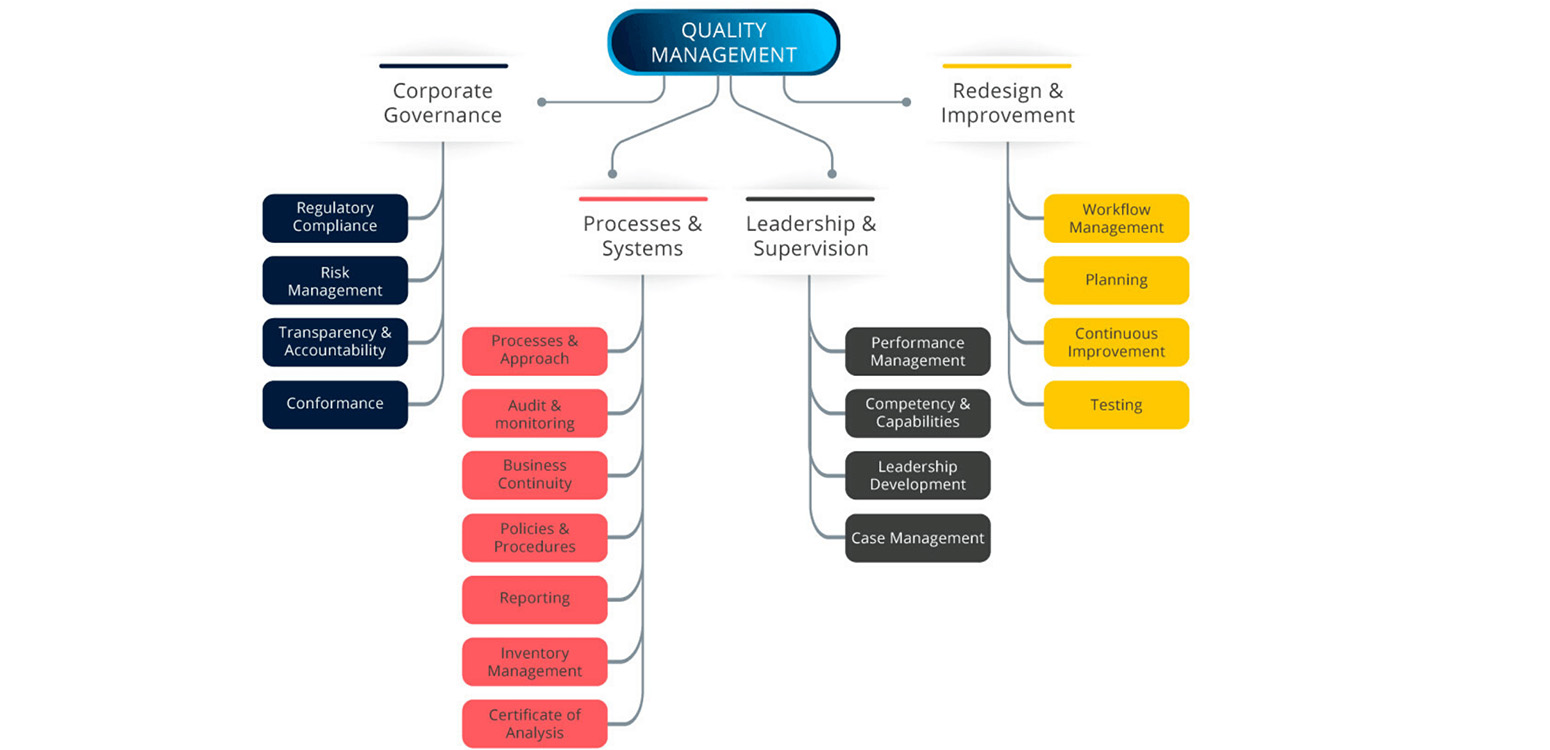
Pharmaceutical Quality Control Improving Methods Systems Xcelpros

Pdf Withdrawn Importance Of Quality Risk Management In Pharmaceutical Quality Systems Recent Trends And Harmonization

New Book On Pharmaceutical Quality Systems Eca Academy

Pharmaceutical Quality Management System Quality Manual 1 Manuals And User Guides Site

Pharmaceutical Quality System Pqs Ispe International Society For Pharmaceutical Engineering

Veeva Expands Into Quality Management Systems Pharmaceutical Commerce
The 5 Best Pharmaceutical Quality Systems Changing Life Sciences In

Drug Quality Fda Officials Discuss Perceptions Raps

Ich Q10 Pharmaceutical Quality Systems An Opportunity For Us All Paperpicks Leading Content Syndication And Distribution Platform

Qms Systems Offers Qms Consulting Services For Manifacturing And Pharma Companies That Realize It Is In Their Best Pharma Companies Management Pharmaceutical

A New Approach To The Pharmaceutical Quality System Acorn Regulatory

Industry 4 0 Innovation And Pharmaceutical Quality Control Mckinsey
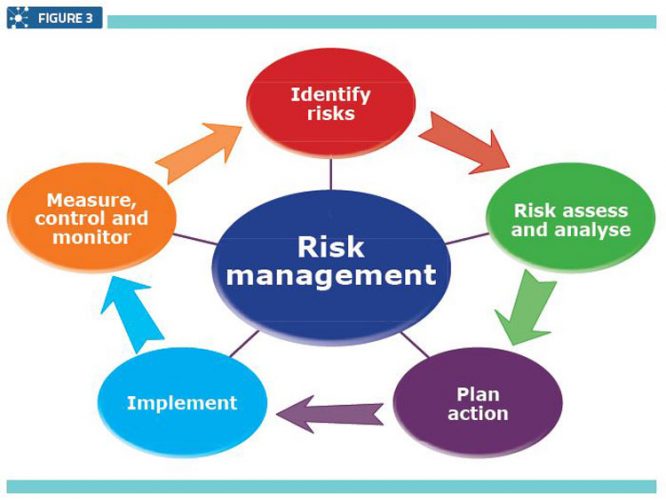
Quality Assurance Pharmaceutical Quality Systems In Making Medicines



